Is Your Mouthful of Metal Healthy?
New FDA guidance says silver fillings may cause health problems for some
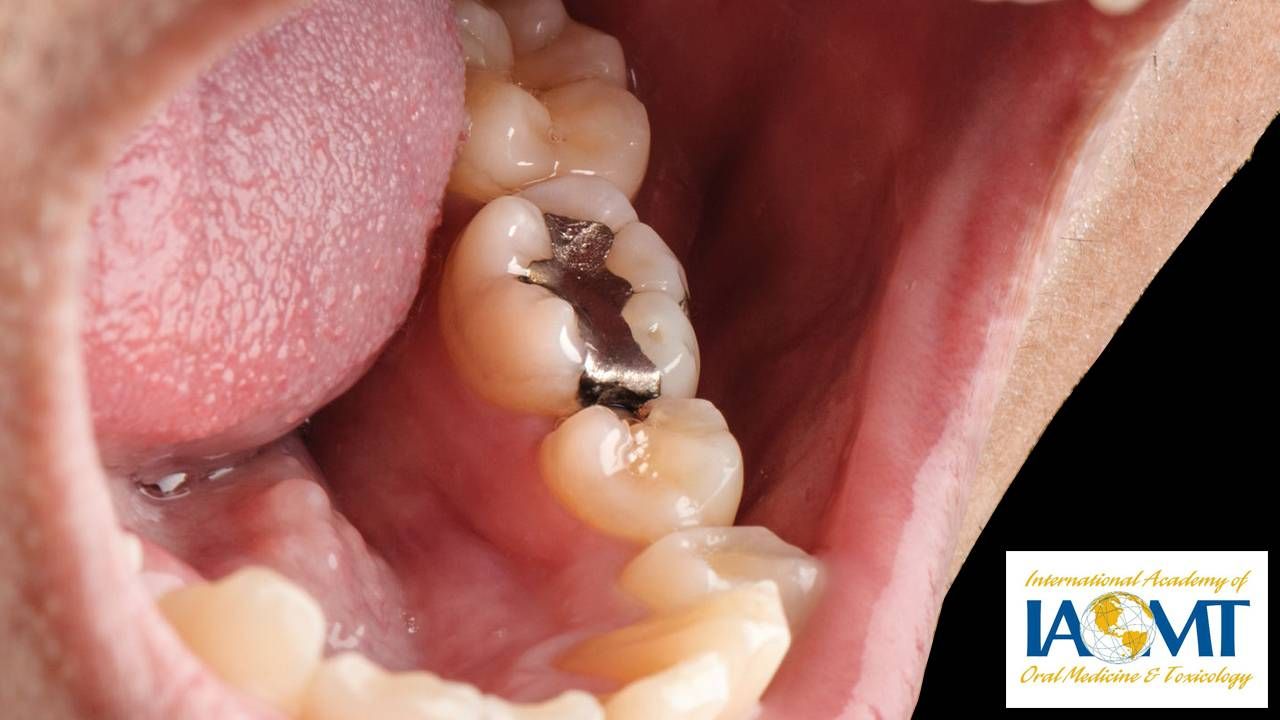
Julio Fernandez has had only four cavities, ever. For the first two, treated when he was young, his dentist used fillings that contained mercury, those silver fillings that have become less popular than the resin-based or ''white'' fillings.
Over the years, Fernandez, now 46 and an international trade professional in Washington, D.C., was aware of the ongoing discussion about whether mercury fillings are safe to leave in or should be removed. Every time he got routine dental care, he asked his dentists about them.
"For years, they would look at them and always say, 'They are fine, they are not showing any cracks. What we advise is, unless they are falling apart that you do not touch them,'" Fernandez says.
The FDA is not recommending that anyone remove or replace existing amalgam filling in good condition unless considered medically necessary.
But then, last summer, when he went in for his routine checkup, his dentist said they had started to crack. The dentist advised removal of the mercury fillings and replacement with a resin-based filling.
FDA Outlines Groups at Higher Risk
Recently, the U.S. Food and Drug Administration (FDA) also weighed in on mercury fillings. In updated guidance issued Sept. 24, the FDA said that the silver fillings, also known as dental amalgam, may cause health problems for some people in high-risk groups. It recommends those who may be at higher risk use non-mercury alternatives, such as the composite resins.
The FDA says groups at higher risk for potential health effects from mercury fillings include:
- Pregnant women and their developing fetuses
- Women planning to get pregnant
- Nursing women and their newborns and infants
- Children, especially those younger than age 6 years
- People with pre-existing neurological diseases, such as multiple sclerosis, Alzheimer's or Parkinson's disease
- People with impaired kidney function
- People with a known allergy to mercury or other components of dental amalgam
The FDA is not recommending that anyone remove or replace existing amalgam filling in good condition unless considered medically necessary. Doing so without medical need, the FDA says, can result in potential loss of healthy tooth structure as the filling is removed, as well as exposure to mercury vapor.
Why the new guidance? The FDA issued it after a public hearing last November. Some experts and members of the public at that meeting expressed concern about the cumulative effect of mercury vapor exposure not only from fillings but other sources, such as the environment.
The Details of Amalgam
Dental amalgam is a mixture that includes mercury, silver tin and copper. At high levels, mercury exposure (from many sources, including environmental ones such as coal burning or mining) can have toxic effects on the nervous, digestive and immune systems as well as the lungs, kidney, skin and eyes, the World Health Organization says.
A filling made of mercury does release a small amount of mercury vapor over time, the FDA says. These low levels are generally not harmful when inhaled but some high-risk people may have issues, according to the FDA. The amount of vapor released from a filling can depend on how old it is and a person's habits, such as grinding of teeth.
What the Research Says
In 2016, researchers from the University of Georgia's College of Public Health analyzed data from nearly 15,000 people, looking at the amount of mercury in their blood and the number of fillings they had. Those with more than eight surfaces of mercury filling did have the highest level of mercury in their blood compared with those who had no fillings or fewer than eight. However, the researchers did not study whether those with more than eight fillings had more health issues.
"They should immediately stop the use of mercury in human beings. Mercury fillings are a two-hundred-year-old mistake."
Since the FDA guidance was issued, Cleveland Clinic weighed in, concluding the fillings are generally safe.
However, other research suggests that susceptibility to mercury toxicity differs among individuals — which appears to support the new FDA guidance — and that efforts to phase down or eliminate the use of mercury fillings are wise.
The ADA Weighs In
In response to the new FDA guidance, the American Dental Association (ADA) released its own statement supporting the guidance. It also agreed with the FDA recommendation that existing amalgam fillings in good condition should not be removed or replaced unless it is viewed as medically necessary by a health care professional.
Deciding on a filling material is an individual decision, the ADA says. "The key here is that the decision has to be made between the dentist and the patient as to what is the right material for this person," says Matthew Messina, a spokesperson for the ADA and director of the dental clinic at The Ohio State University in Columbus.
In recent years, he says, dentists have been doing far fewer amalgam fillings. "Probably seventy percent are not amalgam," Messina says, ''because we have restorative materials [such as composite resins] that look more like teeth. The move away from amalgam has nothing to do with safety and everything to do with aesthetics."
For some people and for some teeth, mercury fillings might be the better option, Messina says. "It is a very forgiving material, and you can place amalgam in an area where it's hard to keep dry, such as in a molar up against the cheek."

If a mercury filling starts to crack, as they did for Fernandez, that's a valid reason to remove them, Messina says.
The fillings are not a risk factor for getting conditions such as Alzheimer's and multiple sclerosis, the ADA notes, even though the FDA urges those who already have those conditions to exercise caution about mercury fillings.
The World Health Organization recommends a ''phase-down'' of the use of mercury, including its use in dental fillings.
One Group Calling for No More Mercury Fillings
The FDA guidance is overdue and does not go far enough, says David Kennedy, past president and now a spokesperson for the International Academy of Oral Medicine and Toxicology (IAOMT), a professional group that has been demanding more stringent protection from dental mercury for more than three decades. The group formed in 1984 and the debate about the safety of the fillings started even before that.
While Kennedy says he is glad the FDA is starting to move forward, it is not enough. "They should immediately stop the use of mercury in human beings," he says. "Mercury fillings are a two-hundred-year-old mistake."
How to Remove Mercury Fillings
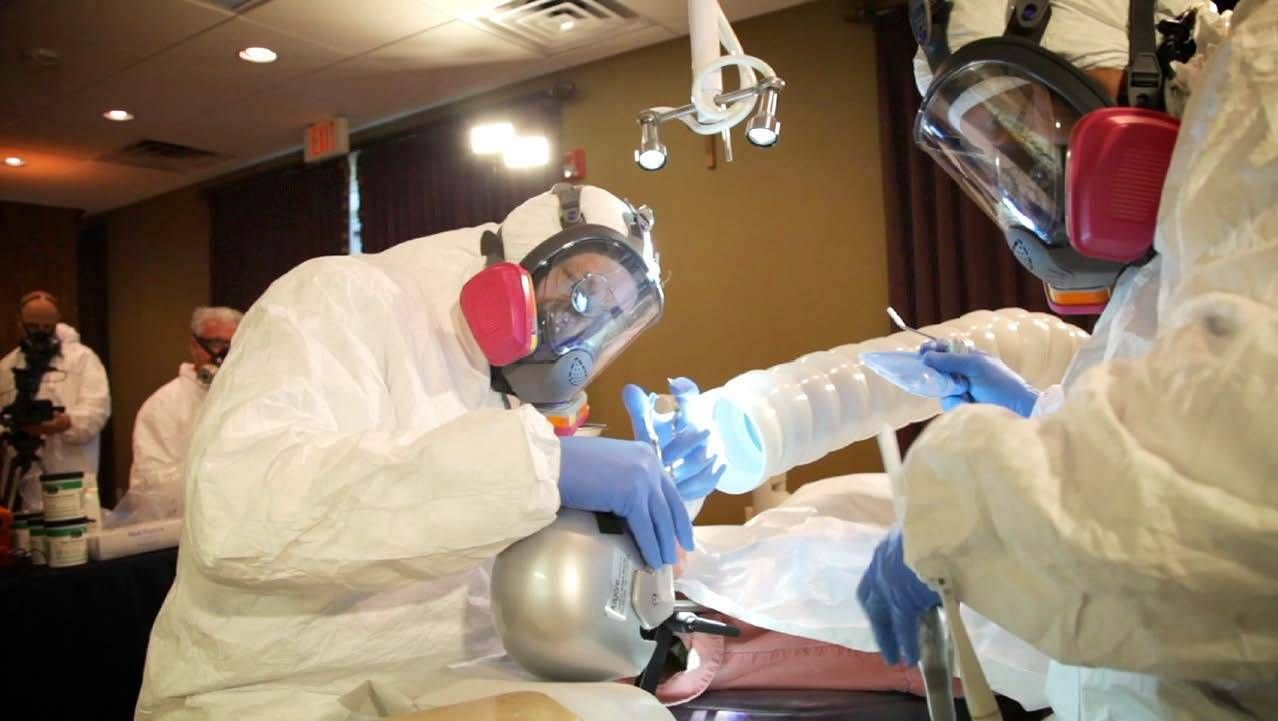
According to Kennedy, those who decide to have the mercury fillings removed — because they are cracked or just to rid their mouth of mercury — should go to a dentist who follows a protocol suggested by his organization, which includes protecting the patient's airway and taking other measures.

According to his organization, numerous symptoms are related to exposure to mercury fillings, including headaches, insomnia, mood swings and fatigue. He says people affected by symptoms due to the mercury fillings often don't connect the two.
Will dental plans pay for removal of mercury fillings and replacement with other materials? Dental plans vary in coverage, but removing them solely for aesthetic reasons is not likely to be covered.
Typically, the composite fillings cost more than the mercury fillings, so it follows that the insurance co-pay is more for composite than amalgam. One popular health plan in Southern California, for instance, charges a co-pay of $40 and up for each silver filling, depending on how many tooth surfaces are involved, and $62 or more for resin, again depending on how many surfaces are involved.
Fernandez had his dentist replace the cracked fillings with composite and says he is doing fine. "I never had any pain after the procedure," says Fernandez.

